
NMPA on IVD Equipment and Hemodialyzer
NMPA published two Q&A on IVD equipment and hemodialyzer respectively, addressing two frequently asked issues: Q: When applying for an in vitro diagnostic equipment registration
Register for Upcoming Webinar on DEC. 8 @ 11AM
2026 China NMPA Bluebook is here:

NMPA published two Q&A on IVD equipment and hemodialyzer respectively, addressing two frequently asked issues: Q: When applying for an in vitro diagnostic equipment registration

NMPA issued fourteen guidelines in July. The guidelines cover indications of ophthalmic, orthopedic, gastroenterology and urology, anesthesia, and immunology, etc. Full List of Guidelines General

The unapproved devices can be clinically used in Hainan to collect real world data (RWD) and support its regulatory approval from NMPA. Under the policy,

NMPA granted Fast-Track Status to two medical device manufacturers in July: Innovation approval status: Clinical Chemistry Shenzhen Guiji Chuangan: continuous glucose monitor Priority review status:

NMPA published the draft version of “Clinical Trial Exemptions Catalog for Medical Devices” on July 29, 2020 for feedback. It is an effort to implement

A Periodic Risk Evaluation Report (PRER) is required to submit to NMPA, depending on the initial NMPA registration time, according to Decree No. 1 Administrative

In March 2018, the transformation from CFDA to NMPA, which emerged into State Administration for Market Regulation (SAMR), was to make the linkage between pre-market

NMPA published 14 guidelines in July 2020. The guidelines cover indications of ophthalmic, orthopedic, gastroenterology and urology, anesthesia and immunology, etc. Full List of Guidelines

A Periodic Risk Evaluation Report is required to submit to NMPA, depending on the initial NMPA registration time, according to Decree No. 1 Administrative Measures
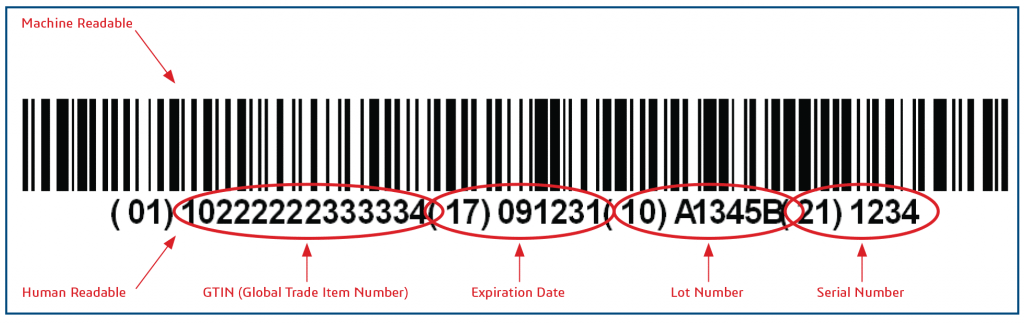
NMPA “Rules for Unique Device Identification (UDI) System” Issued on August 27, 2019 mandates that the China UDI system for the first group of devices

NMPA issued nine orthopedic standards on July 9, 2020 to guide manufacturers for local type testing and regulatory approval. Their implementation date is June 1,

Here’s the June 2020 China NMPA regulatory and clinical affairs news for medical device and IVDs pros. These updates are presented by China Med Device,