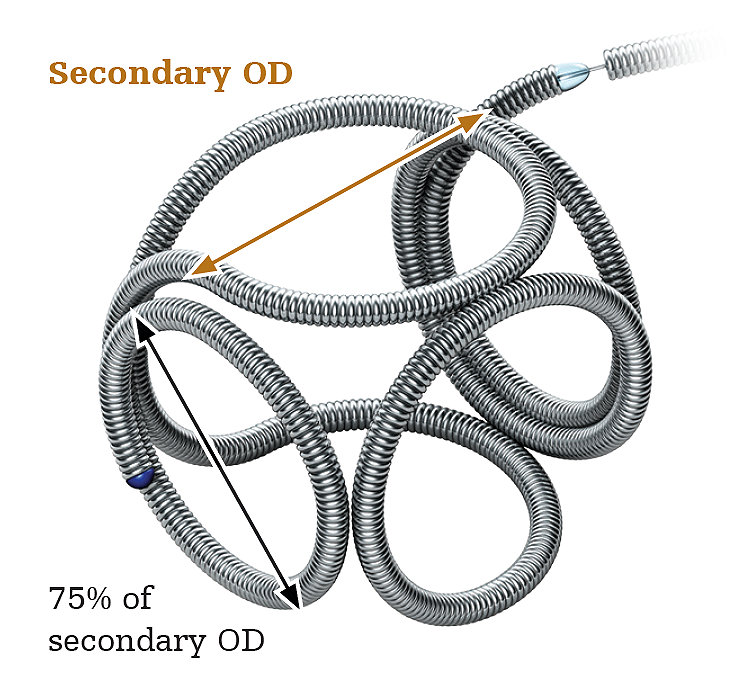
NMPA Clinical Updates: What Overseas Manufacturers Need to Know About the 5 New Class III Devices Guidelines
NMPA published 5 medical devices final guidelines on April 30, 2026. They are aimed to facilitate manufacturers with clarity in clinical evaluation requirements. All five






