
NMPA Roundup May 2026
Here’s the latest China NMPA regulatory and clinical affairs news for medical device and IVDs pros in May 2026. These updates are presented by China
Register for Upcoming Webinar on DEC. 8 @ 11AM
2026 China NMPA Bluebook is here:

Here’s the latest China NMPA regulatory and clinical affairs news for medical device and IVDs pros in May 2026. These updates are presented by China

On March 24, 2026, NMPA published draft versions of registration review key points for 18 medical devices. Once finalized, the documents aim to provide manufacturers
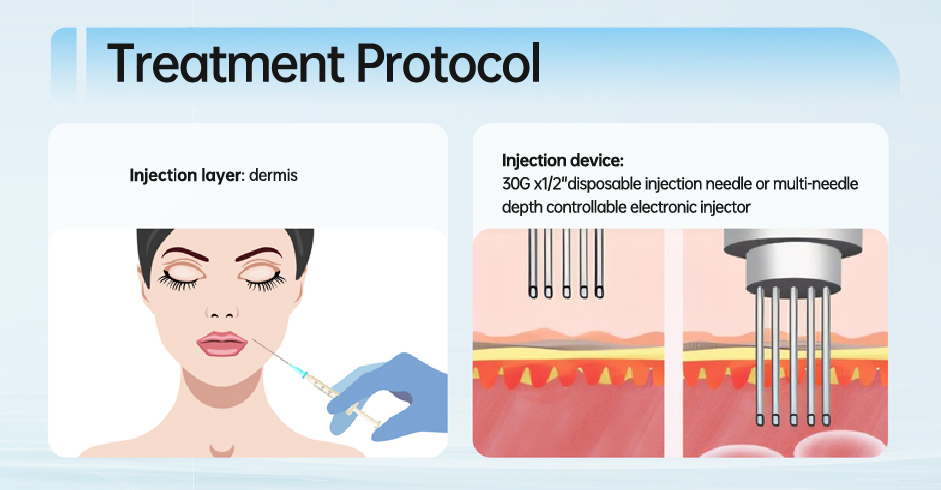
In 2025, polydeoxyribonucleotide (PDRN) has rapidly emerged as one of the most closely watched bioactive materials in the global skincare and medical aesthetics industry. Across

China has applied provisional import tariff rates lower than the most-favored-nation rates on 935 items starting from January 1, 2026, the Customs Tariff Commission of

Here’s the latest China NMPA regulatory and clinical affairs news for medical device and IVDs pros in February 2026. These updates are presented by China

Here’s the latest China regulatory and clinical affairs news for medical device and IVDs pros. These updates are presented by China Med Device, LLC, your

The Shanghai Municipal Medical Products Administration issued the “Provisions on Transferring Medical Devices with Existing Registration Certificates into Shanghai Production” on August 14, 2025. The

The NMPA National Institutes for Food and Drug Control released the draft GB/T 16886.1-2025, “Biological Evaluation of Medical Devices — Part 1: Evaluation and Testing

Here’s the latest China NMPA regulatory and clinical affairs news for medical device and IVDs pros. These updates are presented by China Med Device, LLC,

Here’s the latest China NMPA regulatory and clinical affairs news for medical device and IVDs pros in July 2025. These updates are presented by China
What Devices Received HUD Designation As examples, the following devices, categorized by indications, have been designated HUD status: Indication Device Manufacturers Year of HUD Designated
FDA’s Humanitarian Use Device (HUD) program offers a crucial lifeline to patients with rare conditions who might otherwise be left behind in the march of