
NMPA Finalized Clinical Trial Approval Catalog
As innovative devices and first-of-kind products enter the China market, the NMPA seeks to strengthen the clinical trial approval requirements for certain devices. Not all
Register for Upcoming Webinar on DEC. 8 @ 11AM
2026 China NMPA Bluebook is here:

As innovative devices and first-of-kind products enter the China market, the NMPA seeks to strengthen the clinical trial approval requirements for certain devices. Not all
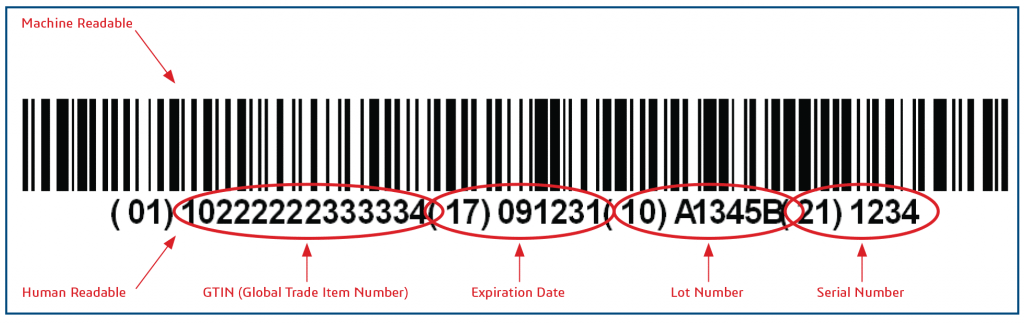
NMPA issued a notice today to delay the UDI implementation date. The first batch of medical devices have changed from October 1st, 2020 to January
Last year, NMPA issued a number of UDI related policies regarding the UDI implementation. There is a UDI database portal (https://udi.nmpa.gov.cn) established which includes sections

NMPA published two Q&A on orthopedic devices, addressing two frequently asked issues: Q: How to determine the mechanical performance indicators when submitting the orthopedic product

NMPA published “Draft Technical Guideline on Medical Device Cybersecurity” on September 8, 2020. Feedbacks need to be submitted to NMPA by October 19, 2020. It

Here’s the latest China regulatory and clinical affairs news for medical device and IVDs pros in August 2020. These updates are presented by China Med

Hainan government published “The System Integration Innovation Reform Plan of Boao Lecheng International Medical Tourism Pilot Zone of Hainan Free Trade Port”, unveiling first-of-its-kind policies

NMPA published seven pathology and clinical chemistry guidelines in August, to direct manufacturers for clinical trial, local type testing and regulatory approval: Clinical trial guideline

NMPA granted Innovation Approvals of two diabetic retinopathy (DR) assistant diagnosis software, to Shenzhen Guiji and Shanghai Yingtong. It is the first time that AI

NMPA launched Unique Device Identification (UDI) Database on its website on August 10, 2020, facilitating the implementation of China UDI system for the first group

NMPA published “Clinical Evaluation Guideline on Medical Magnetic Resonance Imaging (MRI) System Comparing the Similar Device (draft)” on August 13, 2020. The 19-page document provides

Here’s the latest NMPA regulatory and clinical affairs news for medical device and IVDs pros for July 2020. These updates are presented by China Med