
A Chinese Patient Receives a 3D-Printed Knee Joint Implant – The World’s First
According to a local news report, an 84-year-old man in China recently became the first patient in the world to receive a 3D-printed tantalum knee
Register for Upcoming Webinar on DEC. 8 @ 11AM
2026 China NMPA Bluebook is here:

According to a local news report, an 84-year-old man in China recently became the first patient in the world to receive a 3D-printed tantalum knee

Highlights of CFDA’s Plan To Implement Review and Approval System Reform and Encourage Drug and Medical Device Innovation in China In October 2017, China’s

Ortho Clinical Diagnostics announced that its ORTHO VISION Platform of fully automated blood analyzers for low-, mid- and high-volume transfusion medicine laboratories has received approval
China’s Point of Care Testing industry: A billion-dollar market and growing 20-30% annually The rapid growth of the Point of Care Testing Industry and the obvious
China’s In Vitro Diagnosis (IVD) Market is an opportunity for U.S. companies to take their product offering overseas. Here is a current overview of the

EYE TECH CARE, a French medical device company, has received approval from CFDA to begin marketing its EyeOP1® glaucoma treatment product in China. The first
China Med Device cordially invites you to our latest regulatory event in Boston on December 10th, 2017 and the opportunity to meet face to face

The 2017 China BioMed Innovation and Investment Conference (2017 CBIIC) was held in Suzhou China lately. It was co-hosted by Securities Association of China (SAC),

CryoLife, a US-based medical device and tissue processing company focused on cardiac and vascular surgery, announced that enrollment has started in the company’s BioGlue clinical

CFDA just released a Solicitation for Public Comments (SPC) for its proposed amendment of the Medical Device Regulations (MDR), as mandated by a recent order
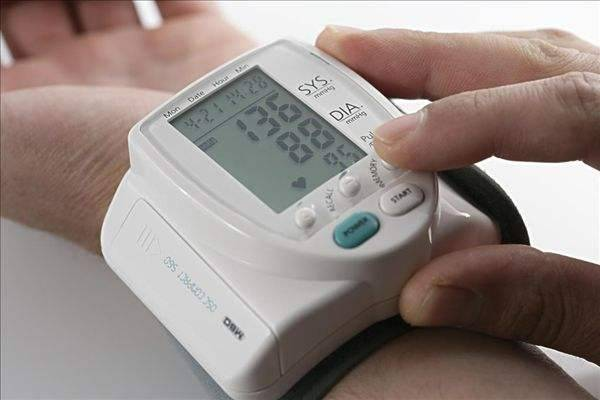
According to a recent market study conducted in China, over a third of Chinese adults are now suffering from high blood pressure. To make matters
On October 8th, China State Council released guidelines for Further Reform Registration Reviewing and Approval to encourage medical device/drug innovation for comments. The guidelines contain