China Medical Device Regulations – An Overview
China National Medical Products Administration regulates medical devices and pharmaceutical products across China. The NMPA was also known as China Food and Drug Administration in
Register for Upcoming Webinar on DEC. 8 @ 11AM
2026 China NMPA Bluebook is here:

China National Medical Products Administration regulates medical devices and pharmaceutical products across China. The NMPA was also known as China Food and Drug Administration in

On March 1st, 2019, Grace Palma, CEO of China Med Device, is interviewed by BioWorld MedTech about the revised registration guidelines for medical devices. With revised

NMPA announced the “2019 Medical Device Registration Guidelines Revision Plan” today (February 25), in which 86 medical devices, IVDs and documentation methods are included. The

Monthly CFDA News Roundup covers government announcement, policies, standards, guidelines, QA/recall/AE, and new approvals in medical device and IVD in China. It is edited by

NMPA (CFDA) published two groups of overseas inspection results on Jan 18 and Jan 30. 17 overseas medical device and IVD manufacturers were cited, including

NORTH ANDOVER, Mass. – January 31, 2019 – ChinaMed Device, LLC (CMD), a leader in helping medtech/IVD companies with RA, QA and CRO in China,

Do you need assistance in NMPA (CFDA) consultation before initiating your registration? In the areas where it is not clearly defined, it is better to

On January 3, 2019, NMPA (CFDA) issued Medical Device Master Files Filing Process (draft) for feedback. Feedbacks need to be submitted by Feb 4, 2019.

NMPA (CFDA) announced “2019 Medical Device Industry Standard Revision Draft Plan” for public comments on December 11, 2018 to medical device/ IVD regulations in order

Monthly CFDA News Roundup covers government announcement, policies, standards, guidelines, QA/recall/AE, and new approvals in medical device and IVD in China. It is edited by

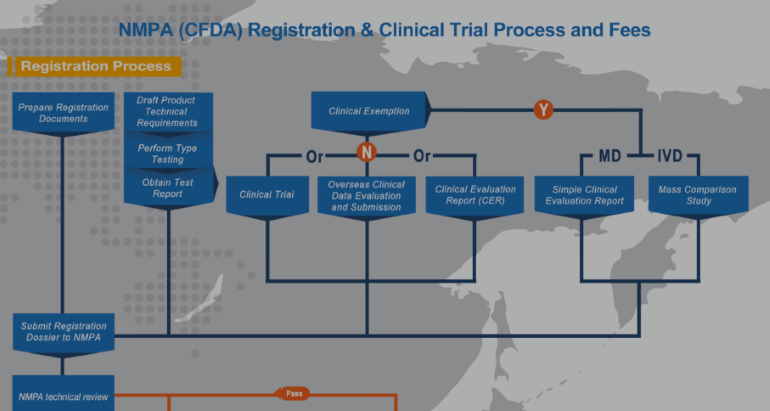
NMPA (CFDA) issued Guideline for Acceptance of Overseas Clinical Trial Data of Medical Devices on January 11th, 2018. A year later, we revisit this important

NMPA (CFDA) issued Guideline for Acceptance of Overseas Clinical Trial Data of Medical Devices on January 11th, 2018. A year later, we revisit this important