China National Medical Products Administration regulates medical devices and pharmaceutical products across China. The NMPA was also known as China Food and Drug Administration in the past. For medical device manufacturers, understanding these regulations is essential. In this post, we give an overview of the NMPA and current China medical device regulations.
About China NMPA
NMPA is an administrative agency of China’s Central Government. It operates under the jurisdiction of the State Administration for Market Regulation. Specifically, NMPA creates, supervises, and implements policies and standards governing medical devices, cosmetics, and drugs. The agency also oversees the registration process for these products, as well as inspections.
As a regulatory body, NMPA has 10 functions:
- Management and supervision of drugs, cosmetics, and medical devices.
- Establishing standards for drugs, cosmetics, and medical devices.
- Managing registration of drugs, cosmetics, and medical devices.
- Handling quality management for all regulated products.
- Risk management of regulated products after listing.
- Managing qualification and registration of licensed pharmacists.
- Inspection and supervision of drugs, cosmetics, and medical devices.
- Handling foreign exchanges and other interactions with international parties.
- Guiding drug supervision and administration departments under government jurisdiction.
- Completing any other assigned tasks.
Accordingly, NMPA is organized into different departments and institutes. In particular, they include the Department of Medical Device Registration, Department of Policy, Law, and Regulation, China’s National Institute for Food and Drug Control, Technology and International Cooperation, and Department of Medical Device Supervision and Administration.
China Medical Device Regulations – Medical Device Classification
The NMPA classifies medical devices into three categories: Class I, Class II, and Class III. Specifically,
Class I – Medical devices for which routine administration can ensure safety for users and the effectiveness of the device.
Class II – Medical devices that can only be safe and effective with further control in addition to routine administration.
Class III – Medical devices that are implanted into the patient’s body, pose a threat to the patient’s health, or provide sustenance or life support.
Medical Device Registration
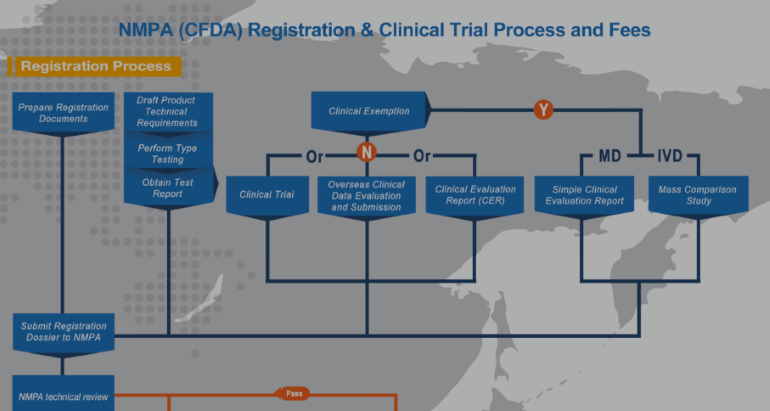
All medical devices must be registered with the NMPA. Furthermore, overseas device manufacturers should follow a specific registration process. Specifically, an overseas device company must submit product samples to test with the NMPA. In addition, all included product information, packaging, and labels, etc. need to be translated into simplified Chinese. For a Class I devices, you just need to do simple product filing to NMPA. However, for Class II and Class III medical devices, the manufacturing company must meet all the requirements in the latest regulation, guidelines, and standards.
Other Relevant China NMPA Medical Device Regulations
The NMPA uses a Unique Device Identification (UDI) system to improve the tracking and monitoring of medical devices. This system stores device production dates, models, codes, and expiration dates. Click HERE for the details.
Also, the NMPA prioritizes the development and registration of innovative medical devices. If your product qualifies as an innovative device, you may not even need home country approval. Email info@chinameddevice.com to request further details about the China innovation pathway.
There are many specific China medical device regulations. For example, NMPA medical device clinical trial regulations apply to the manufacturing and sale of medical devices in China. Also, keep in mind that regulations that apply to medical devices in China may change over time. So it is important to review updated regulations to ensure ongoing compliance. Our NMPA newsletters keep you updated with medical device regulations in China. Please click HERE to opt-in.
About ChinaMed Device, LLC
ChinaMed Device, LLC (www.ChinaMedDevice.com) provides regulatory and commercialization turnkey solutions for medical device, IVD, CDx and combination products in China. As a certified NMPA (CFDA) legal agent with offices in Boston and Beijing, we represent manufacturers for the complete product life cycle without their need to create a local entity in China. Our NMPA (CFDA) regulatory services include strategy, registration, local type testing, product technical requirement (PTR), clinical evaluation report (CER), clinical trial, GMP and post-market surveillance (AE, recall, inspection, etc.). Our commercialization services include market research, reimbursement, partnership strategy and distribution qualification.