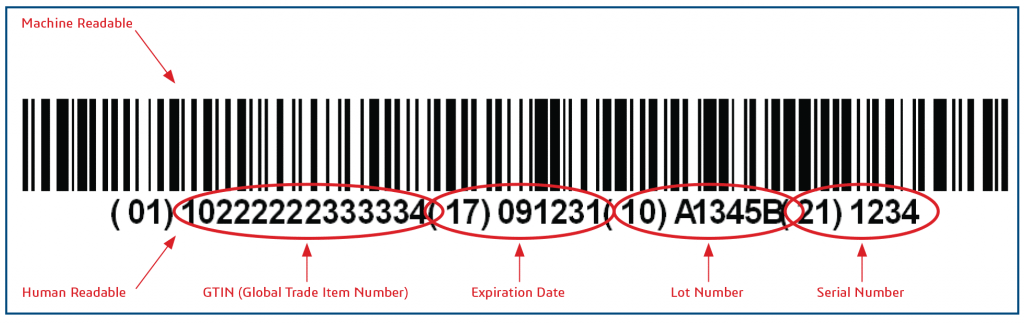
China Medical Device UDI: First Batch Implementing October 1, 2020
NMPA “Rules for Unique Device Identification (UDI) System” Issued on August 27, 2019 mandates that the China UDI system for the first group of devices
Register for Upcoming Webinar on DEC. 8 @ 11AM
2026 China NMPA Bluebook is here:

NMPA “Rules for Unique Device Identification (UDI) System” Issued on August 27, 2019 mandates that the China UDI system for the first group of devices

NMPA issued nine orthopedic standards on July 9, 2020 to guide manufacturers for local type testing and regulatory approval. Their implementation date is June 1,

Here’s the June 2020 China NMPA regulatory and clinical affairs news for medical device and IVDs pros. These updates are presented by China Med Device,

As innovative devices and first-of-kind products enter China market, NMPA seeks to strengthen the clinical trial approval requirements for certain devices. Not all of them

NMPA granted fast-track channel status to two domestic AI-infused software on June 29, 2020: Innovation Status Zhiyuan Huitu: Assistance software for fundus diagnosis Priority Review

NMPA granted Innovation Approval to Edwards Lifesciences’ Transcatheter Aortic Valve System on June 8. It is the 14th device obtained such approval in 2020. Balloon
Thursday, July 9, 2020 | 12:00 pm – 1:10 pm ET Registration is Closed China has been increasing its surveillance on post market monitoring with

NMPA issued four orthopedic guidelines on June 9, 2020 to guide manufacturers for local type testing and regulatory approval. Registration Guideline on 3D printed acetabular

“Regulations on the Management of Imported Medical Devices Urgently Needed in the Hainan Pioneer Zone” was published by Hainan government on June 2, 2020. This

NMPA issued the finalized version of 2020 Medical Device Industry Standards Revision Plan on May 19, in which 86 medical devices and IVDs are affected.

Here’s the latest China NMPA regulatory and clinical affairs news for medical device and IVDs pros from May 2020. These updates are presented by China

China’s Center for Medical Device Evaluation (CMDE), a division of NMPA announced its preparation of “Technical Review Guideline of Clinical Trial for Co-development of CDx