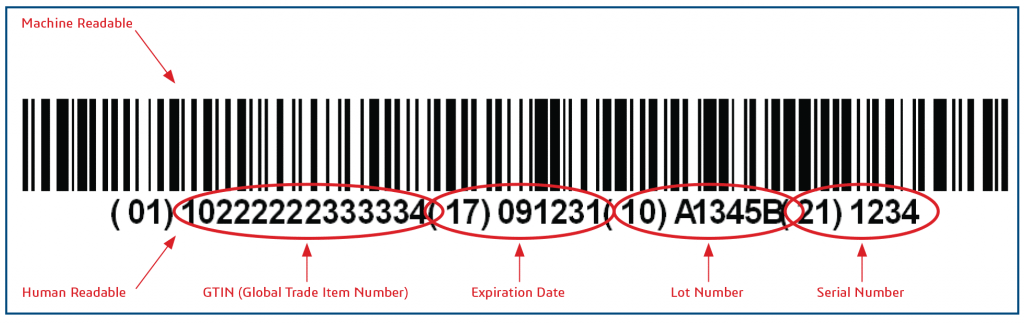
China NMPA Delayed UDI Implementation Date to Jan 1st, 2021
NMPA issued a notice today to delay the UDI implementation date. The first batch of medical devices have changed from October 1st, 2020 to January
Register for Upcoming Webinar on DEC. 8 @ 11AM
2026 China NMPA Bluebook is here:

NMPA issued a notice today to delay the UDI implementation date. The first batch of medical devices have changed from October 1st, 2020 to January
Last year, NMPA issued a number of UDI related policies regarding the UDI implementation. There is a UDI database portal (https://udi.nmpa.gov.cn) established which includes sections
Thursday, July 9, 2020 | 12:00 pm – 1:10 pm ET Registration is Closed China has been increasing its surveillance on post market monitoring with

Amid the novel Coronavirus (COVID-19) outbreak, we deliver the Coronavirus Special Roundup to give you insights on the measures China NMPA has taken to handle
Wednesday, March 18, 2020 | 12:00 pm – 12:45 pm ETIn response to the Novel Coronavirus (COVID-19) outbreak, China regulatory agencies issued over 70 Emergency Approvals

While the Novel Coronavirus (COVID-19) has caused a tragic loss of life in China, the crisis demonstrates the flexibility of the country’s health agency. In

You may find it complicated and confusing when trying to navigate through China’s medical device classification, registration and clinical trials process. In addition, to make

Pic: China Premier Li Keqiang visited the Hainan International Medical Tourism Pilot Zone on Mar 25, 2019. NMPA issued the “Guideline on Real World Data

NMPA announced Recall Notices for 16 imported medical devices in November. Class I recall (the most serious): Edwards Lifesciences: Aortic perfusion cannula Class II and

Striving to meet good manufacturing practices (GMP) for medical devices in China can be challenging. The following article will provide answers to the most common

NMPA published five guidelines for 3D printed devices from August to October 2019: Registration Guideline on 3D Printed Implantable Bone, Joint and Oral Hard Tissue

International Medical Device Regulators Forum (IMDRF) Committee Members meeting was held on September 17-19, in which China NMPA achieved two milestones. The commitments, on post-market-surveillance