
NMPA: Four Important Guidelines Ask for Feedback
The NMPA issued four draft guidelines today on September 28, 2022, soliciting public opinions. Your NMPA guidelines feedback needs to be submitted by October 14,
Register for Upcoming Webinar on DEC. 8 @ 11AM
2026 China NMPA Bluebook is here:

The NMPA issued four draft guidelines today on September 28, 2022, soliciting public opinions. Your NMPA guidelines feedback needs to be submitted by October 14,

The NMPA released the draft version of “Registration Guideline for Radio Frequency Aesthetic Devices” on August 26. 2022 for feedback. Opinions need to be submitted

Here are the latest China NMPA Regulatory and Clinical Affairs news for medical device and IVDs pros for July 2022. These updates are presented by

The NMPA released the “Guideline for Fundamental Principles Compliance of Medical Device Safety and Performance”, elaborating the risk-based approach for medical device registration review. Article

Here’s the latest China NMPA regulatory and clinical affairs news for medical device and IVDs pros in June 2022. These updates are presented by China

The NMPA released 55 revised or newly established medical device standards on May 18, 2022, with indications ranging from cardiovascular, plastic surgery, oncology, clinical chemistry
Hainan Boao special zone allows overseas unapproved medical devices, IVDs, drugs in China to be used. In addition to the overseas medical products being sold
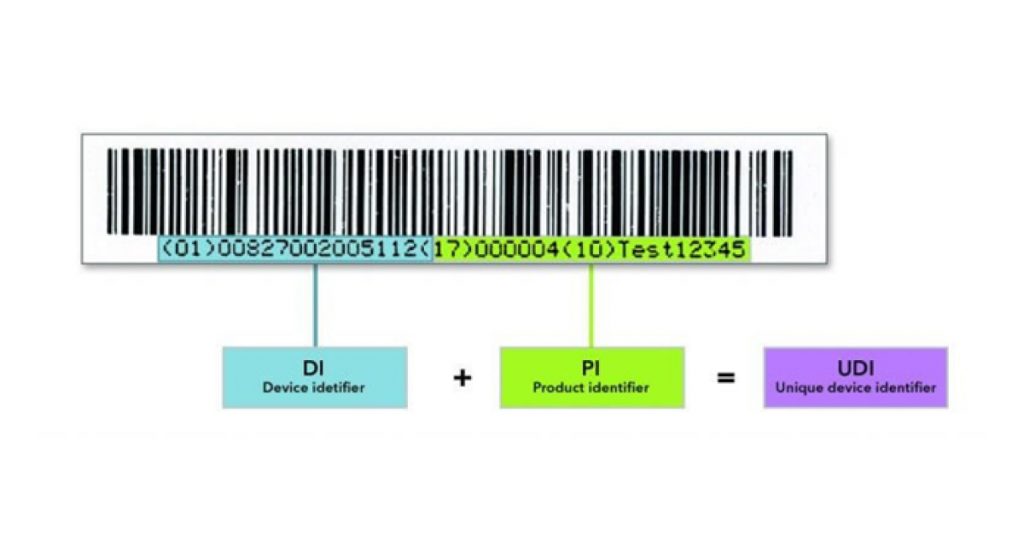
Unique Device Identification (UDI) will be implemented with all Class III devices and IVDs, effective from June 1, 2022. It was announced by NMPA at

Here are the latest China regulatory and clinical affairs news pieces for medical devices and IVDs in February 2022. These updates are presented by China

The NMPA issued the “Guideline on Artificial Intelligence Medical Devices (Draft)” on June 4 for feedback. The document is to direct manufacturers on how define,

China Med Device, LLC joined the NMPA Session on interpreting the “Notice of Medical Device Submission Material Requirements and Certificate Format” held on December 16,

The NMPA released 19 industry standards on December 10, 2021, with indications ranging from cardiovascular, plastic surgery, oncology, clinical chemistry and immunology, etc. The standards