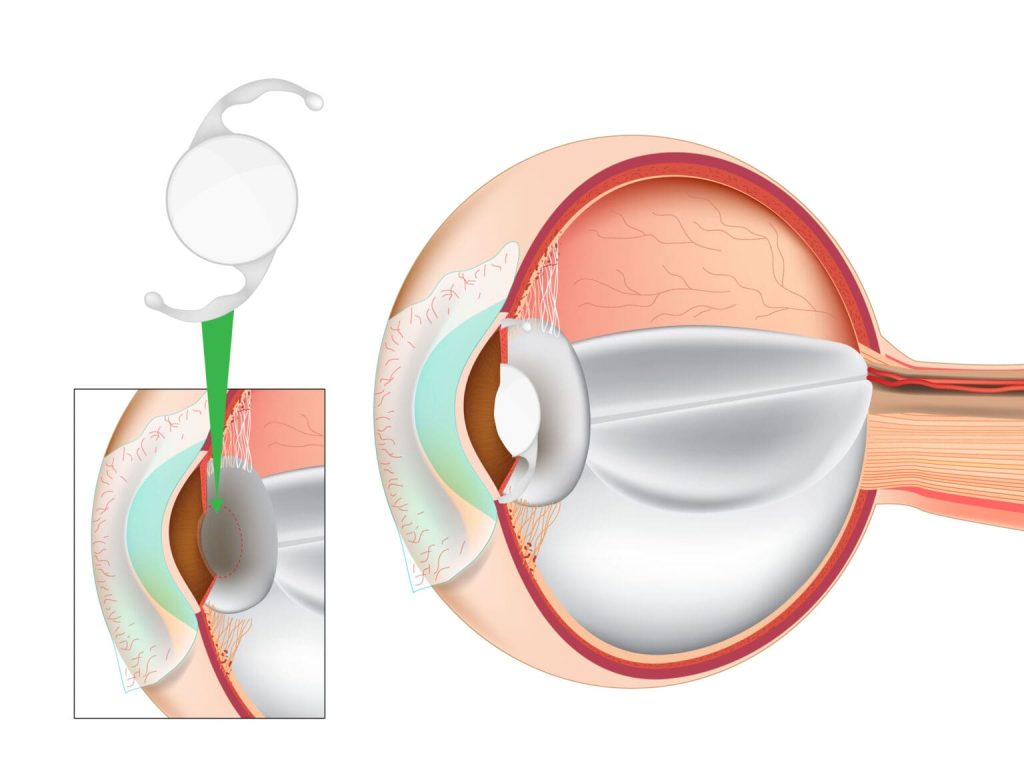
Clinical Applications of Optical Coherence Tomography (OCT) in Ophthalmology: with MIOCT and Ocular Injection in Focus (I)
Optical Coherence Tomography (OCT) has become an indispensable imaging tool in ophthalmology, offering high-resolution, non-invasive, real-time cross-sectional visualization of ocular tissues. Since its introduction in