Dry eye disease (DED) is a prevalent and chronic ocular condition characterized by tear film instability, ocular discomfort, and visual disturbances. With the increasing use of digital screens, aging populations, and changing environmental conditions, the incidence of DED continues to rise globally. While traditional treatments such as artificial tears and anti-inflammatory agents offer symptomatic relief, they often suffer from poor patient adherence and long-term safety concerns. In recent years, punctal occlusion therapies—particularly those utilizing innovative biodegradable materials like polyvinylpyrrolidone (PVP), poly(lactic-co-glycolic acid) (PLGA), and hyaluronic acid (HA)—have emerged as promising alternatives that physically preserve tear volume and improve ocular surface health. This article explores the science, clinical value, regulatory status, and future directions of these novel therapeutic modalities.
I. Punctal Occlusion Therapy: From Concept to Clinical Implementation
The lacrimal drainage system begins at the puncta—small openings in the eyelids that serve as the entry point for tear outflow. Punctal occlusion works by physically blocking these ducts, thereby reducing tear drainage, increasing tear retention, and stabilizing the tear film. This intervention can significantly alleviate the symptoms and signs of moderate to severe dry eye disease.
Compared to irreversible thermal cauterization, modern occlusion methods using removable or biodegradable materials represent a minimally invasive, reversible, and customizable approach to managing DED. These treatments are especially valuable for patients who do not respond adequately to topical medications or prefer less frequent intervention.
Core Mechanism and Material Innovation
1. Physical Occlusion without Pharmacologic Action
The primary therapeutic mechanism of punctal plugs and gels is mechanical. By physically occupying the canalicular space, these devices prevent tear drainage without relying on pharmacologic, immunologic, or metabolic pathways. The effectiveness of the treatment hinges on the material’s biocompatibility, structural conformity to the canaliculus, and the duration of in situ stability.
2. Breakthroughs in Biocompatible Materials
PVP-Based Plugs
One widely adopted product employs semi-soft polyvinylpyrrolidone (PVP) hydrogel, which expands upon contact with tears. This hydrophilic behavior allows the plug to conform to the shape of the canaliculus, ensuring a snug and effective seal. The material demonstrates excellent biocompatibility, structural integrity, and can be flushed out when necessary, offering a balance between efficacy and reversibility.

PLGA-Based Biodegradable Plugs
PLGA, a copolymer of polylactic acid and glycolic acid, has long been used in biodegradable medical devices. When fashioned into punctal plugs, PLGA offers a controlled degradation timeline—typically spanning several months. The breakdown products are gradually absorbed by surrounding tissues, eliminating the need for removal procedures. Available in multiple sizes and formulations, PLGA plugs allow clinicians to tailor treatment to individual patient anatomy and disease severity.

Hyaluronic Acid-Based Canalicular Gels
HA-based injectable gels represent a newer class of punctal occlusion tools. These formulations combine crosslinked sodium hyaluronate with sterile water to form a prefilled injectable hydrogel. Upon delivery into the canaliculus, the HA forms a hydrated, viscous, crosslinked matrix that blocks tear outflow. The hydrophilic and lubricating properties of HA provide additional comfort. Crosslinking extends the duration of occlusion—typically lasting around six months—after which the gel biodegrades naturally.
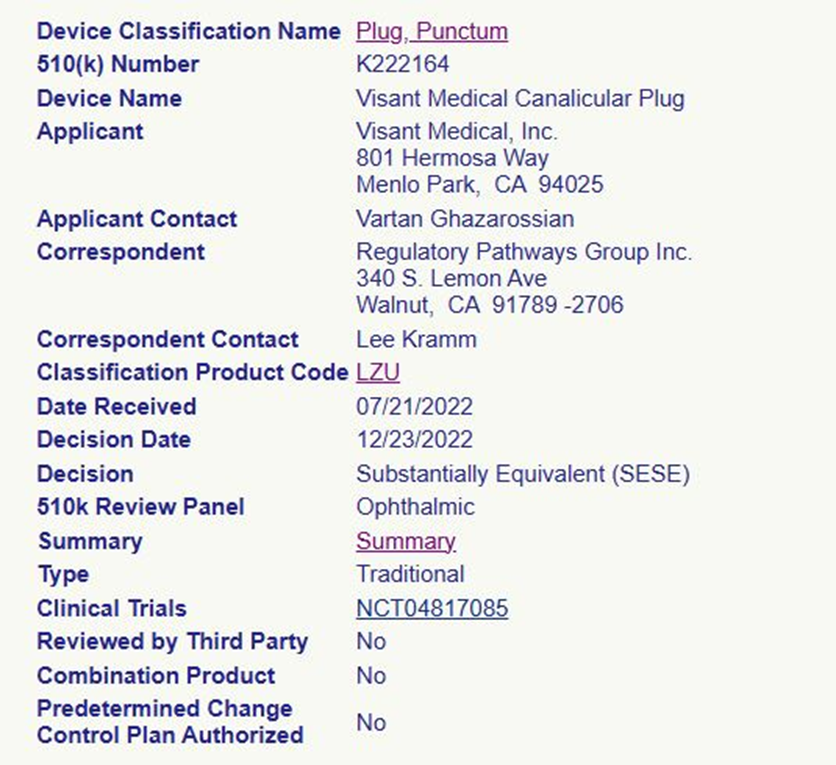
Nordic Pharma launched a new generation of punctal occlusion device, LACRIFILL Canalicular Gel, in 2024, which utilizes a proprietary cross-linked hyaluronic acid for occlusive treatment. It is biocompatible, long-lasting, and comfortable for patients. Moreover, it is reversible and repeatable—if a patient’s symptoms begin to return, the treatment can be administered again.
Based on clinical research results, the U.S. FDA approved its use. Studies have shown that the gel is safe for patients and can effectively retain natural tears in the eye for up to six months.
LACRIFILL is typically implanted in an outpatient setting, and the entire process takes only a few seconds. It can be administered under a slit lamp or with the aid of magnifying loupes while the patient is lying back in a chair, requiring only local anesthesia. Preparing LACRIFILL is quick and easy: simply attach the chosen cannula and ensure it is firmly connected. Draw 0.1 mL of LACRIFILL for pre-filling; stop when gel appears at the cannula tip. Inject 0.1 mL of the gel into the punctum and stop when reflux is observed. As with a traditional punctal plug, insert the cannula tip into the punctum.
In most cases, dilation is not required, nor is it necessary to occlude all four puncta. It is recommended to begin with the lower punctum. The implantation procedure can be performed by optometrists, ophthalmologists, or physician assistants.
For dry eye specialists and cataract surgeons, LACRIFILL serves as either a final adjunctive therapy or a standalone treatment option, addressing unmet needs while also providing a valuable source of revenue.

LACRIFILL is applied to the punctum using the inserter or cannula selected by the surgeon.
FDA 510K clearance of LACRIFILL

