
Importation Halted for KARL STORZ’s High-frequency Surgery System Due to QMS Issues
The NMPA issued an inspection result on March 31, 2026 for German manufacturer KARL STORZ’s High-frequency Surgery System. NMPA organized a remote inspection of KARL
Register for Upcoming Webinar on DEC. 8 @ 11AM
2026 China NMPA Bluebook is here:

The NMPA issued an inspection result on March 31, 2026 for German manufacturer KARL STORZ’s High-frequency Surgery System. NMPA organized a remote inspection of KARL

NMPA announced the “2026 National Inspection Plan for Medical Devices” on April 8, 2026. The Plan asks provincial NMPA offices and testing centers to conduct

Here’s the latest China regulatory and clinical affairs news for medical device and IVDs pros in April 2026. These updates are presented by China Med

On March 24, 2026, NMPA published draft versions of registration review key points for 18 medical devices. Once finalized, the documents aim to provide manufacturers

On March 17, 2026, the NMPA released 26 medical device industry standards, including the revised YY/T 1406—2026: Guidance on the Application of GB/T 42062 to

The NMPA, together with National Health Commission and National Healthcare Security Administration, issued the “Advancing UDI Implementation for Subsequent Device Categories” and “UDI Implementation under

The NMPA Center for Medical Device Evaluation (CMDE) issued the “Pre-Review of Clinical Trial Protocols for Innovative Medical Devices” on March 13, 2026. This initiative

The NMPA approved Shanghai Lixiao’s thermal vapor therapy equipment and the single-use prostate thermal vapor therapy device on February 28, 2026. The thermal vapor therapy

Here’s the latest China NMPA regulatory and clinical affairs news for medical device and IVDs pros in March 2026. These updates are presented by China
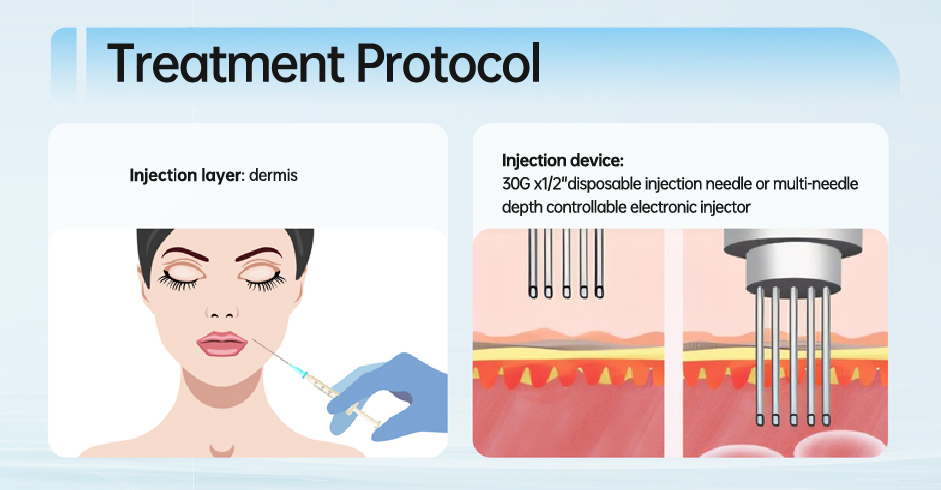
In 2025, polydeoxyribonucleotide (PDRN) has rapidly emerged as one of the most closely watched bioactive materials in the global skincare and medical aesthetics industry. Across

The National Institutes for Food and Drug Control of NMPA released the draft GB/T 16886.1-2025, “Biological Evaluation of Medical Devices — Part 1: Evaluation and

NMPA published the “Guideline for Registration of Mobile Medical Devices” on May 7, 2025, replacing the 2017 version. This updated guideline is to facilitate manufacturers