
NMPA: 55 Class III Devices impacted by 2025 Guidelines Revisions Plan
NMPA published the “2025 Medical Device Guidelines Revisions Plan” on May 7, 2025, in which 55 guidelines for Class III and 55 for Class II
Register for Upcoming Webinar on DEC. 8 @ 11AM
2026 China NMPA Bluebook is here:

NMPA published the “2025 Medical Device Guidelines Revisions Plan” on May 7, 2025, in which 55 guidelines for Class III and 55 for Class II

NMPA published the “Clinical Evaluation Exempt Catalog for Medical Devices” on May 13, 2025, which newly-includes 27 medical devices, including 7 Class III and 20

Here’s the latest China regulatory and clinical affairs news for medical device and IVDs pros. These updates are presented by China Med Device, LLC, your

NMPA published the “Implementation Rules for the Priority Approval Review of Medical Devices” on April 18, 2025. The document is based on the “Medical Devices

In recent years, active devices based on laser, ultrasound, and radiofrequency technologies have gained significant popularity in the field of aesthetic medicine due to their
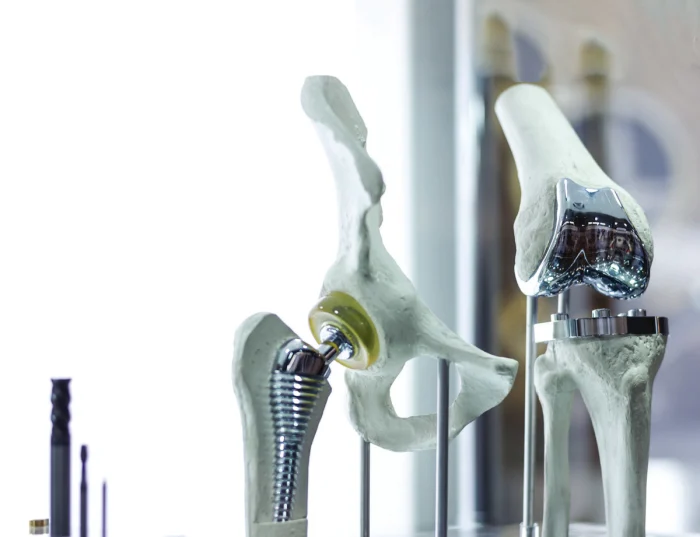
NMPA Regulations for AM Medical Devices NMPA places high importance on the regulation of 3D-printed medical devices. Custom AM implants typically fall under Class III

Additive Manufacturing (AM), or 3D printing technology, is triggering a profound technological revolution in the global orthopedic field. With its personalized, precise, and rapid manufacturing

NMPA published the “Implementation Rules for the Special Review of Innovative Medical Devices” on April 18, 2025. The document is based on the “Innovative Medical

NMPA published the seven IVD reagents guidelines on March 31, 2025. They are aimed to facilitate manufacturers with more product-specific guidance and clarity in registration

Cataracts remain one of the leading causes of vision impairment worldwide, particularly affecting the aging population. In China, statistics show that 27.45% of people over

Here’s the latest China regulatory and clinical affairs news for medical device and IVDs pros in April 2025. These updates are presented by China Med

The NMPA released a notice “Announcement on Further Adjusting and Optimizing Matters Related to the Production of Imported Medical Devices in China” on 18 March,