
Roundup – NMPA News October 2020
Here are the latest NMPA regulatory and clinical affairs news articles in October 2020 for medical devices and IVDs pros. These updates are presented by
Register for Upcoming Webinar on DEC. 8 @ 11AM
2026 China NMPA Bluebook is here:

Here are the latest NMPA regulatory and clinical affairs news articles in October 2020 for medical devices and IVDs pros. These updates are presented by

The NMPA released the IMDRF (International Medical Device Regulators Forum) draft document of “PMCF (Post-Market Clinical Follow-Up) Studies” on October 22, 2020. The proposed document

Electronic signature, certificate and documents have gained popularity due to higher efficiency and availability overseas. Submission Material Electronic Signature NMPA issued a release called “Matters

With an announcement on October 20, 2020, the NMPA reclassified 29 cancer biomarker-related reagents from Class III to Class II. Additionally, they modified its intended

Thirty four industry standards for medical devices were issued by the NMPA on October 22, 2020. These were put in place to guide manufacturers for

The NMPA issued a notice called “the Establishment of Medical Robotics Standardization Agency” on September 27. This is announcing the regulatory preparation for medical robotics.

NMPA granted priority review designation to CooperVision and Biomerieux on September 30, and gave its reason as below: Soft hydrophilic contact lens, by CooperVision, on

CER (Clinical Evaluation Report) plays a vital role in the clinical pathway in China and predicate-comparison is the key element of CER. NMPA published Guideline

NMPA published the draft document of National Medical Device Quality Inspection Sampling Procedure on October 13, 2020 for feedback. Feedback needs to be submitted by
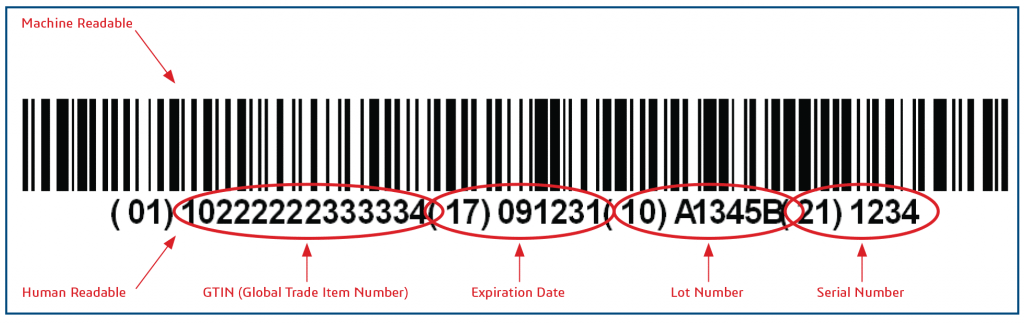
NMPA launched Unique Device Identification (UDI) Database on its website on August 10, 2020, facilitating the implementation of China UDI system for the first group

Here’s the latest China NMPA regulatory and clinical affairs news in September 2020 for medical device and IVDs pros. These updates are presented by China

NMPA published the final notice “Issues regarding Approved Imported Medical Devices Manufactured by Chinese Domestic Enterprises” on September 25, 2020. The draft version was released